Signifor May be Safe, Effective Option to Treat Cushing’s Long-term, Study Indicates
Written by |
Signifor (pasireotide), an approved therapy for Cushing’s disease, has shown promise as a long-term treatment for some patients with the disease, according to results of an extension study of a Phase 3 clinical trial (NCT00434148).
These findings were published recently in the study, “Long-term treatment of Cushing’s disease with pasireotide: 5-year results from an open-label extension study of a Phase III trial,” in the journal Endocrine.
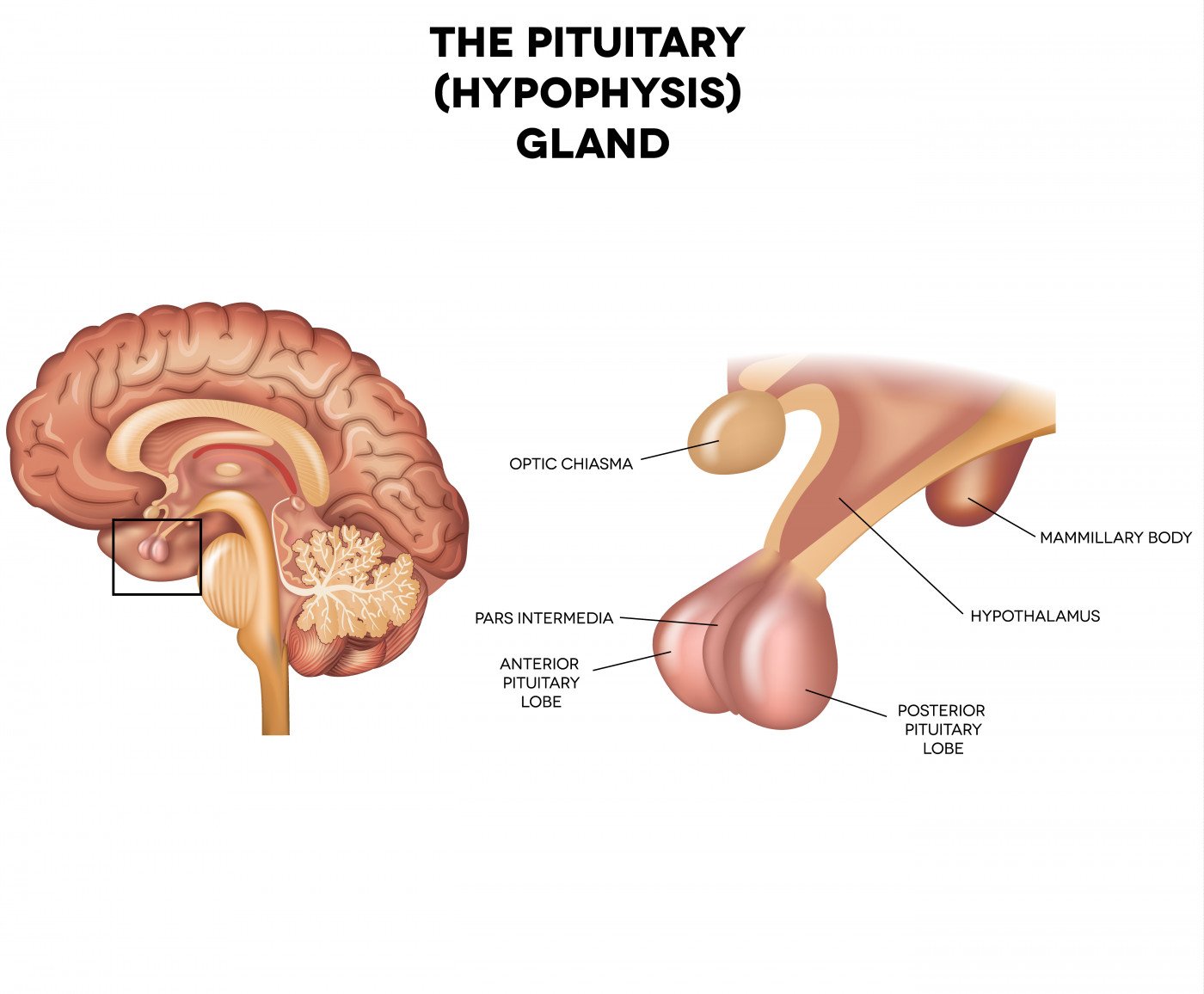
Cushing’s disease is marked by increased secretion of the adrenocorticotropic hormone (ACTH), or cortisol, a feature called hypercortisolism. This often is caused by the growth of a mass of cells in the anterior pituitary gland, the structure that produces ACTH in the body, causing its excessive production.
Treating hypercortisolism in Cushing’s disease patients who do not respond properly to surgery requires permanent medication. For this reason, drug therapies must have long-term effectiveness and safety.
“Earlier reports from a large Phase III study … showed that pasireotide reduced urinary free cortisol (UFC) levels and provided clinical benefit for up to 24 months in patients with Cushing’s disease,” researchers wrote. However, there is limited information on the effectiveness and safety of this treatment beyond 24 months of treatment.
To address this matter, researchers investigated the long-term effects of Signifor in 162 adult patients with persistent or recurrent Cushing’s disease. Patients were randomly assigned to receive one of two doses of subcutaneous Signifor (600 µg or 900 µg, twice daily).
Those considered to benefit from the treatment after 12 months were allowed to continue receiving Signifor in the extension phase. Also, if the benefit persisted and Signifor was well-tolerated, patients could continue treatment until the drug became commercially available.
Of the initial group of patients, 16 received Signifor treatment for five years. These patients presented a median change in the mean UFC levels of -82.6 percent at month 12 and -81.8 percent at month 60, compared to the study’s start. At month 60, 11 of these patients had a mean UFC below or equal to the upper limit of normal UFC.
The benefits induced by Signifor were sustained over the five-year period, with a safety profile similar to that seen after one year of treatment. Most patients (15) had an adverse side effect associated with increased blood sugar (hyperglycemia). Side effects related to hyperglycemia, including bradycardia (slow heart beat), gallbladder/biliary tract, and liver safety were most likely to first appear after six months of treatment. Importantly, the severity of adverse effects did not worsen with time.
Together, these results indicate that treatment with Signifor may be a valuable option for the long-term treatment of Cushing’s disease.
“In the longest-running prospective clinical trial of a medical therapy for Cushing’s disease to date, [Signifor] treatment reduced median mUFC and improved disease-related clinical signs,” researchers wrote. “These beneficial effects were sustained for up to 5 years in the subgroup of patients who remained on treatment. No new safety signals were identified during long-term treatment.”